About

Hi, I am a half-brazilian half-lithuanian evolutionary biologist. I investigate how different evolutionary forces like sexual and natural have shaped morphological changes through time. In my research I focus on reproductive traits, which evolve so fast and so uniquely in many animal groups of internal fertilization. I like to approach these questions in a multi-scale framework: from single individuals (ontogeny), to populations and phylogenies.
I started in science studying the taxonomy of stink bugs during my undergrad course. I soon realized there were tons of genera whose general morphology were relatively static, but their genitalia were astonishingly diverse. I immersed myself in the literature of genital evolution and realized that this was actually a widespread phenomenon across animals. I learned that genital traits were actually kind of special in evolutionary terms. However, there were many more open questions than answers in this realm.
The study of genital evolution, to which I've been devoting my time since my Masters, has helped to answer fascinating and broad questions in evolutionary biology. Why some traits are much more complex than others? Why some features evolve extremely fast, while others are virtually unchanged? How come males and females may exhibit so many behavioral and morphological differences, in spite of having more or less the same genome? Which signatures have all these processes left in the genome of animals? To answer these questions, I combine phylogenetic comparative methods, genome-scale data, and morphometric analyses.
In my spare time, I enjoy brewing drinks, playing heavy stuff on the guitar and woodworking.
EDUCATION
- 2018, PhD in Systematics, Animal Taxonomy and Biodiversity
[University of São Paulo]
- 2014, MSc in Ecology and Evolution
[Federal University of São Paulo]
- 2011, BSc in Biological Sciences
[Federal University of São Paulo]

Research
CONNECTING FUNCTION, DEVELOPMENT AND ADAPTATION
There is a fine tune among organs and tissues during morphogenesis that guarantee the correct specification of an organism's parts. This process is controlled by very complex and entangled networks of regulatory genes. Many of these genes participate in the formation of different organs, which makes development a highly pleiotropic and integrated process. Developmental integration is a key property of evolution as it may constrain the availability of morphological variation available for selection to act.
Function is another key component of morphological evolution since selection acts over functional demands. Genitalia in insects are remarkable structures to study the interplay between function and development. This is because insect genitalia are functionally complex, with many parts developing distinct functions, but also developmentally complex, with their parts originating from multiple tissues/segments.
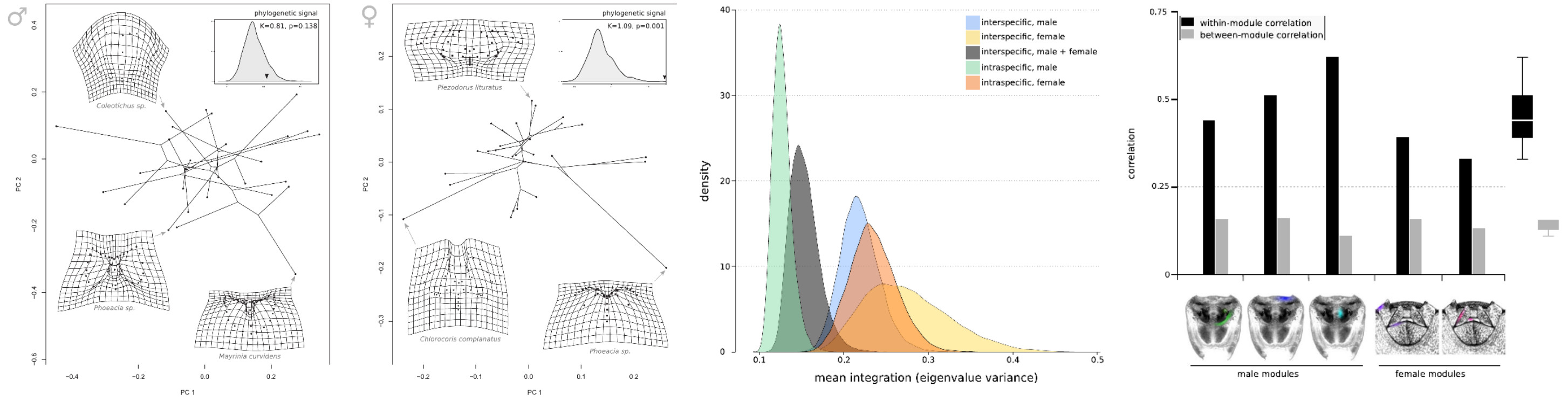
I combine developmental data and functional morphology to understand which of these two factors are more determinant to genital evolution. In a study from 2020 published in Evolution, we showed that development is a constraining factor more relevant for females of stink bugs, as we detected clear developmental modules. In contrast, function is more determinant for the modular organization of male genitalia, as the parts that function together in copule comprise modules. This pattern is consistent within and among species, suggesting an evolutionary conservation of genital modularity.
COMPARATIVE METHODS AND THE TEMPO OF EVOLUTIONARY CHANGE
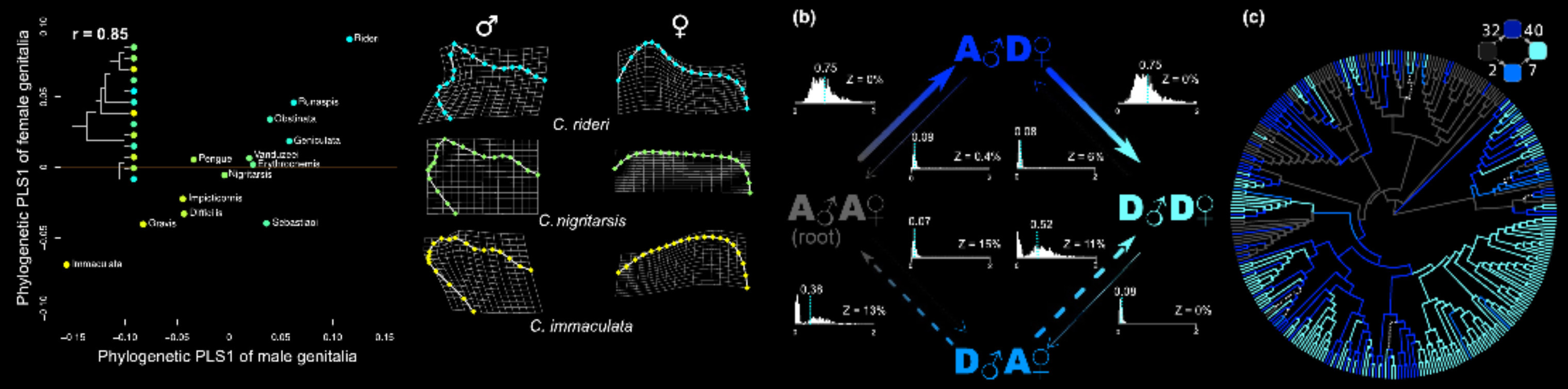
Tempo of evolution refers to the rates of change exhibited by phenotypes and genotypes through time. Combining molecular phylogenies, fossils, and specific statistical methods, it is possible to estimate how fast different traits have changed. The rate of change may give important clues about how evolution have proceeded. For example, really fast evolving traits may have experienced more intense selection comparing to slow traits. In such a context, genitalia are probably the best models to elucidade the mechanisms of accelerated evolution since they are the fastest evolving traits known for insects and many other groups.
Using phylogenetic comparative methods applied to genital traits, I investigate the tempo of genital change and look for signatures of potential underlying processes. For example, along with my collaborators, I showed for the first time that female genitalia may be fastly evolving just like male genitalia, contrary to the common belief in the classic taxonomic literature. In another study, we investigated the time points in which male and female genitalia of beetles have shifted from membranous to rigid (and vice versa). We showed that changes in female genitalia almost always preceeded male changes. This suggests that the coevolution between male and female genitalia may be "female-oriented" in certain cases, and again, that the role of female structures in the course of evolution had been overlooked.
GENOMIC BASES OF PHENOTYPIC EVOLUTION
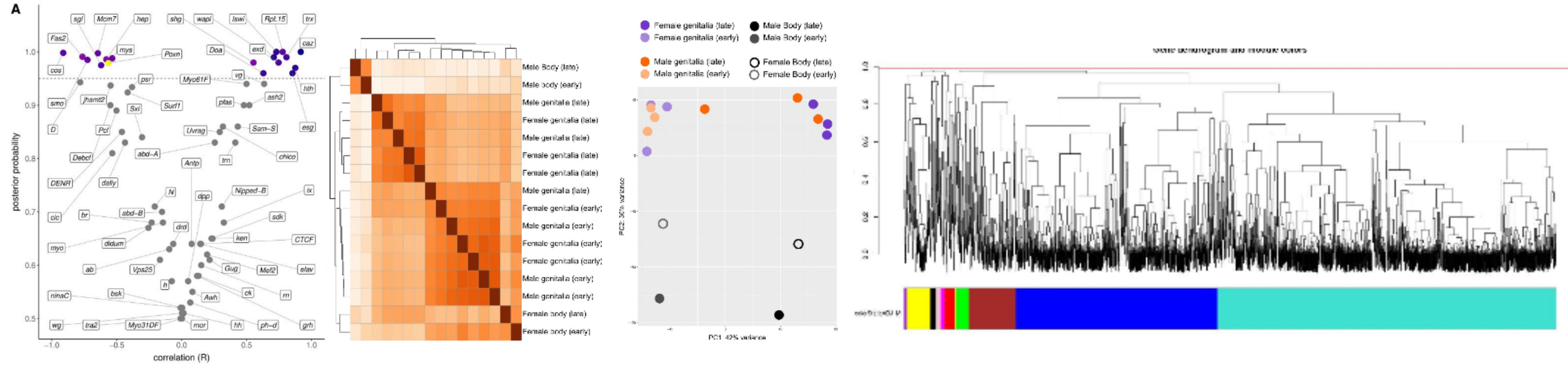
The same genes that regulate development, from the egg to the adult, are the genes that are targets of natural selection and other processes. As a result, we expect that the patterns and processes that govern the evolution of phenotypes will leave specific signatures in the genome of organisms. Understanding the connection between genotype and phenotype, specially in large time scales, is what motivates most of my research nowadays. I have been combining different kinds of genome-scale data like gene expression levels, sequence composition, gene family sizes and others, to study the origins of genital complexity. For now, we have discovered that the macroevolution of genital size has left selection signatures in several genes that regulate different morphogenetic processes, suggesting that genital size per se is a complex trait. But wait for more! We are discovering many cool things regarding what governs genital plasticity and how novel genital traits arise.




